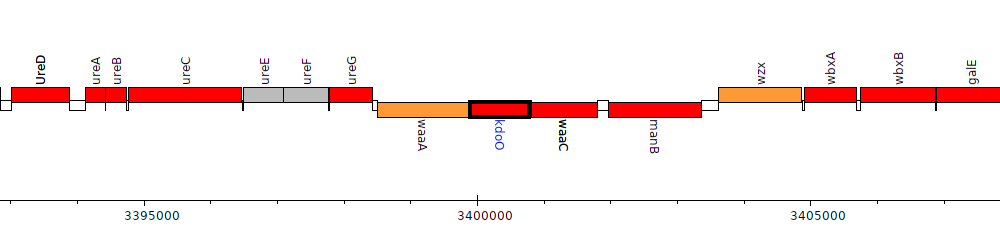
Burkholderia cenocepacia J2315, BCAL3111 (kdoO)
Cytoplasmic
Cytoplasmic Membrane
Periplasmic
Outer Membrane
Extracellular
Unknown
Gene Feature Overview
| Strain |
Burkholderia cenocepacia J2315
GCF_000009485.1|latest |
| Locus Tag |
BCAL3111
|
| NCBI Locus Tag | QU43_RS52285 |
| Name |
kdoO
Synonym: wbxY |
| Replicon | chromosome 1 |
| Genomic location | 3399895 - 3400779 (- strand) |
| Comment | Responsible for Ko formation with Kdo2-lipid A as a substrate |
Cross-References
| RefSeq | WP_006485119.1 |
| GI | 206561457 |
| Entrez | 6934235 |
| GI | 206561457 |
| INSDC | CAR53435.1 |
| NCBI Locus Tag | QU43_RS52285 |
| NCBI Old Locus Tag | BCAL3111 |
| RefSeq | WP_006485119.1,GeneID |
| UniParc | UPI00017B994F |
| UniProtKB Acc | B4ECD2 |
| UniProtKB ID | B4ECD2_BURCJ |
| UniRef100 | UniRef100_B4ECD2 |
| UniRef50 | UniRef50_Q0WGC8 |
| UniRef90 | UniRef90_B4ECD2 |
Product
| Feature Type | CDS |
| Coding Frame | 1 |
| Product Name |
Kdo 3-hydroxylase KdoO
|
| Synonyms |
hypothetical protein |
| Evidence for Translation | |
| Charge (pH 7) | 3.59 |
| Kyte-Doolittle Hydrophobicity Value | -0.222 |
| Molecular Weight (kDa) | 32740.3 |
| Isoelectric Point (pI) | 8.48 |
Subcellular localization
| Individual Mappings | |
| Additional evidence for subcellular localization |
Pathogen Association Analysis
| Results |
Common
Found in both pathogen and nonpathogenic strains
Hits to this gene were found in 28 genera
|
Orthologs/Comparative Genomics
| Burkholderia Ortholog Group |
BG000751 (654 members) |
| Putative Inparalogs | None Found |
Interactions
| STRING database | Search for predicted protein-protein interactions using:
Search term: BCAL3111
Search term: kdoO
Search term: Kdo 3-hydroxylase KdoO
|
Human Homologs
References
|
Reconstitution of O-specific lipopolysaccharide expression in Burkholderia cenocepacia strain J2315, which is associated with transmissible infections in patients with cystic fibrosis.
Ortega X, Hunt TA, Loutet S, Vinion-Dubiel AD, Datta A, Choudhury B, Goldberg JB, Carlson R, Valvano MA
J Bacteriol 2005 Feb;187(4):1324-33
PubMed ID: 15687196
|
|
The LpxL acyltransferase is required for normal growth and penta-acylation of lipid A in Burkholderia cenocepacia.
Fathy Mohamed Y, Hamad M, Ortega XP, Valvano MA
Mol Microbiol 2017 Apr;104(1):144-162
PubMed ID: 28085228
|
|
Dioxygenases in Burkholderia ambifaria and Yersinia pestis that hydroxylate the outer Kdo unit of lipopolysaccharide.
Chung HS, Raetz CR
Proc Natl Acad Sci U S A 2011 Jan 11;108(2):510-5
PubMed ID: 21178073
|
|
Kdo hydroxylase is an inner core assembly enzyme in the Ko-containing lipopolysaccharide biosynthesis.
Chung HS, Yang EG, Hwang D, Lee JE, Guan Z, Raetz CR
Biochem Biophys Res Commun 2014 Sep 26;452(3):789-94
PubMed ID: 25204504
|